Supporting Toxicology Studies
As a GLP accredited company, we undertake histology for preclinical toxicological studies for all species in-line with recognised industry standards.
We specialise in conducting standardised and specialised toxicity study sample assessments and reporting, from the receipt of wet tissue samples to the delivery of fully audited GLP Pathologist reports. Our comprehensive histology services support various preclinical studies, including efficacy, pharmacodynamics (PD), pharmacokinetics (PK), and dose escalation studies.


Our toxicology study services include:
Routine toxicology, oncology studies
Specialised – inhalation, neurotoxicity, immunotoxicity
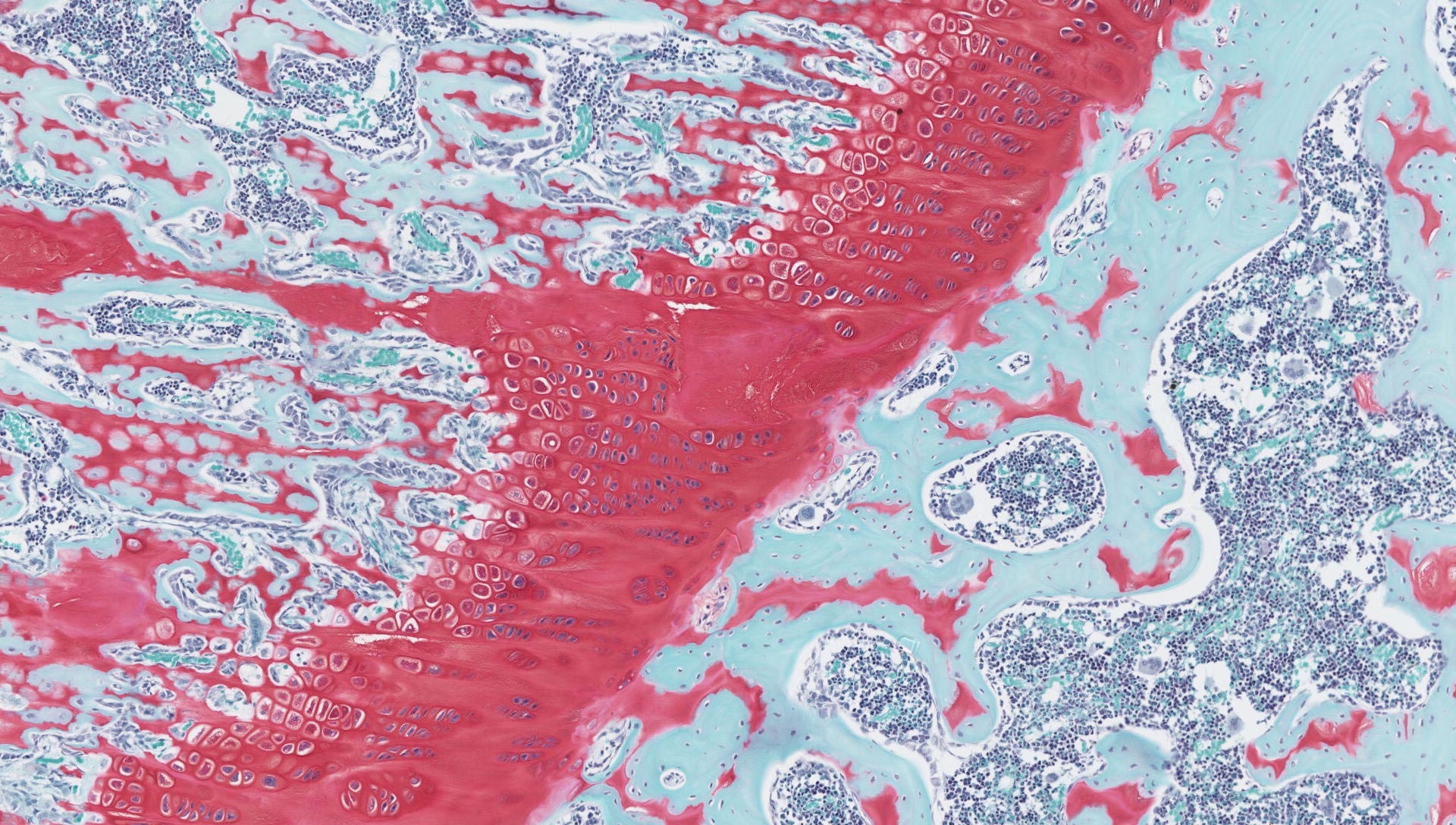
Specialised tissues – bone, nasal turbinates, larynx, etc
Standardised and custom trimming planes, blocking codes and species-specific processing schedules
Decalcified tissues
Megablock soft and hard decalcified tissues
Tissue trackdown and step serial sections
Standard H&E or client specific H&E staining used
Client SOPs and documentation used on request
Meet agreed timelines and fast-track timelines as required
Why Choose HistologiX to Evaluate Your Toxicology Study Tissue Samples?
1
Experienced Scientists
Our scientists carry out standardised and specialised toxicity study sample assessment and reporting, starting from wet tissue sample receipt.
We have optimised protocols for handling all tissue types. Providing optimal processing, trimming and embedding of all tissue types harvested from toxicology studies and disease models.
2
Pathology Review
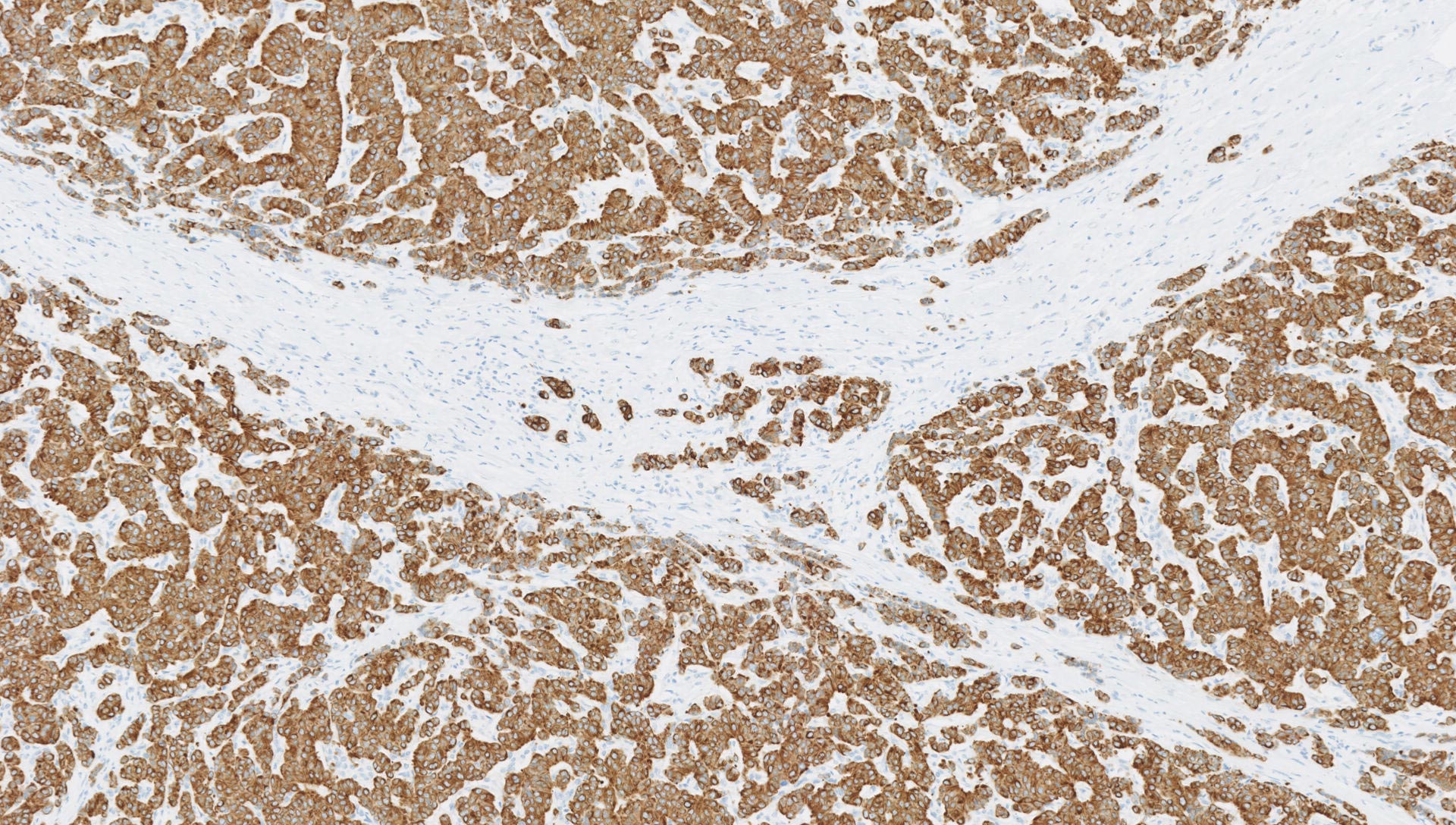
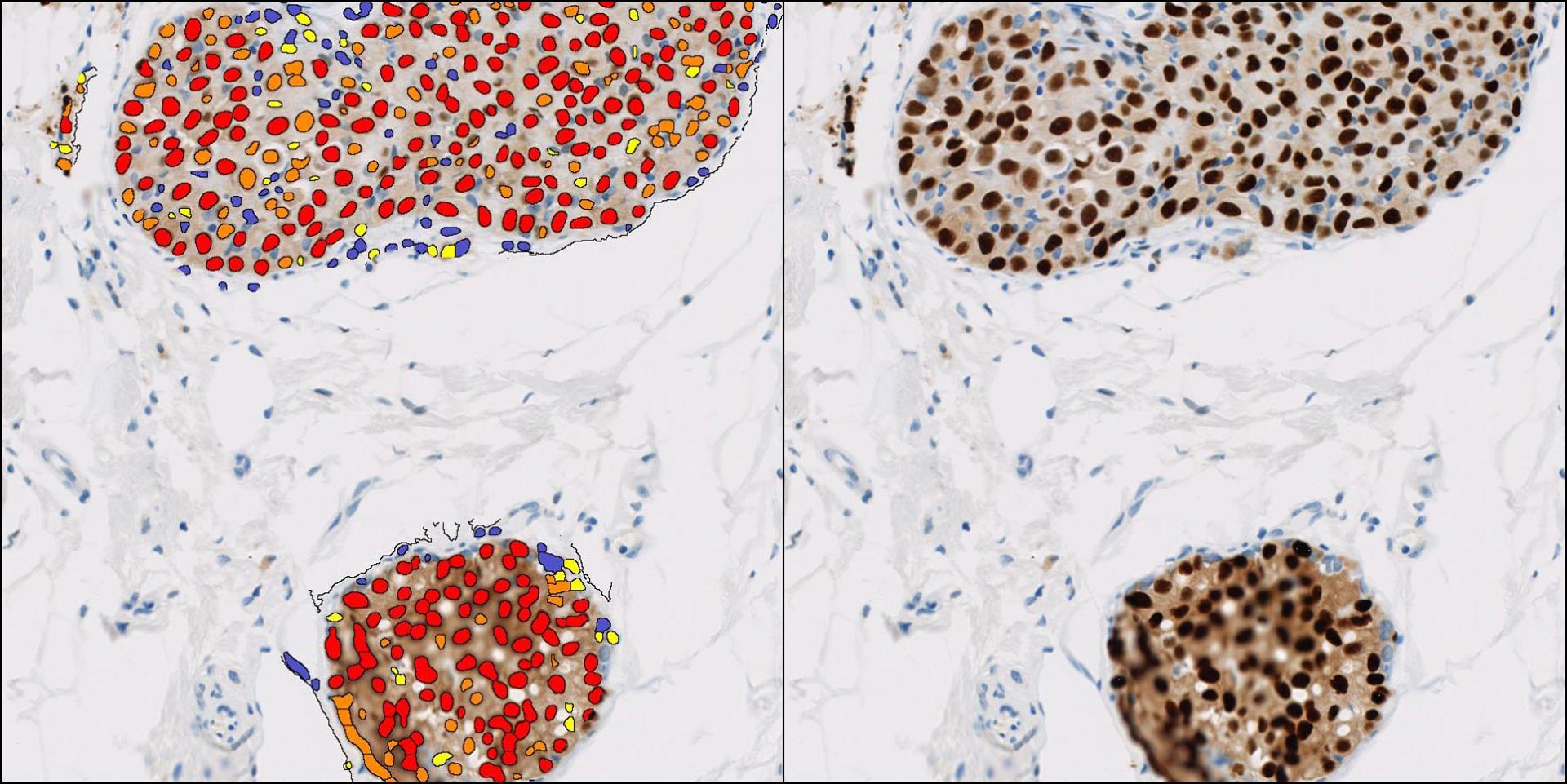
Following H&E staining, physical slides or digital scans are sent for pathology review.
We work with GLP accredited, board certified Veterinary Pathologists for assessment of your preclinical studies and safety assessment for regulatory submissions.
3
Regulatory Compliance
Our services adhere to GLP standards, ensuring the reliability and acceptance of your study data by regulatory authorities.
With the capability to process a range of sample types, whether from biobanks, pre-clinical or clinical studies, we are ideally placed to enable you to get the most out of your projects in accordance with your GxP regulatory requirements.
4
Other Applications
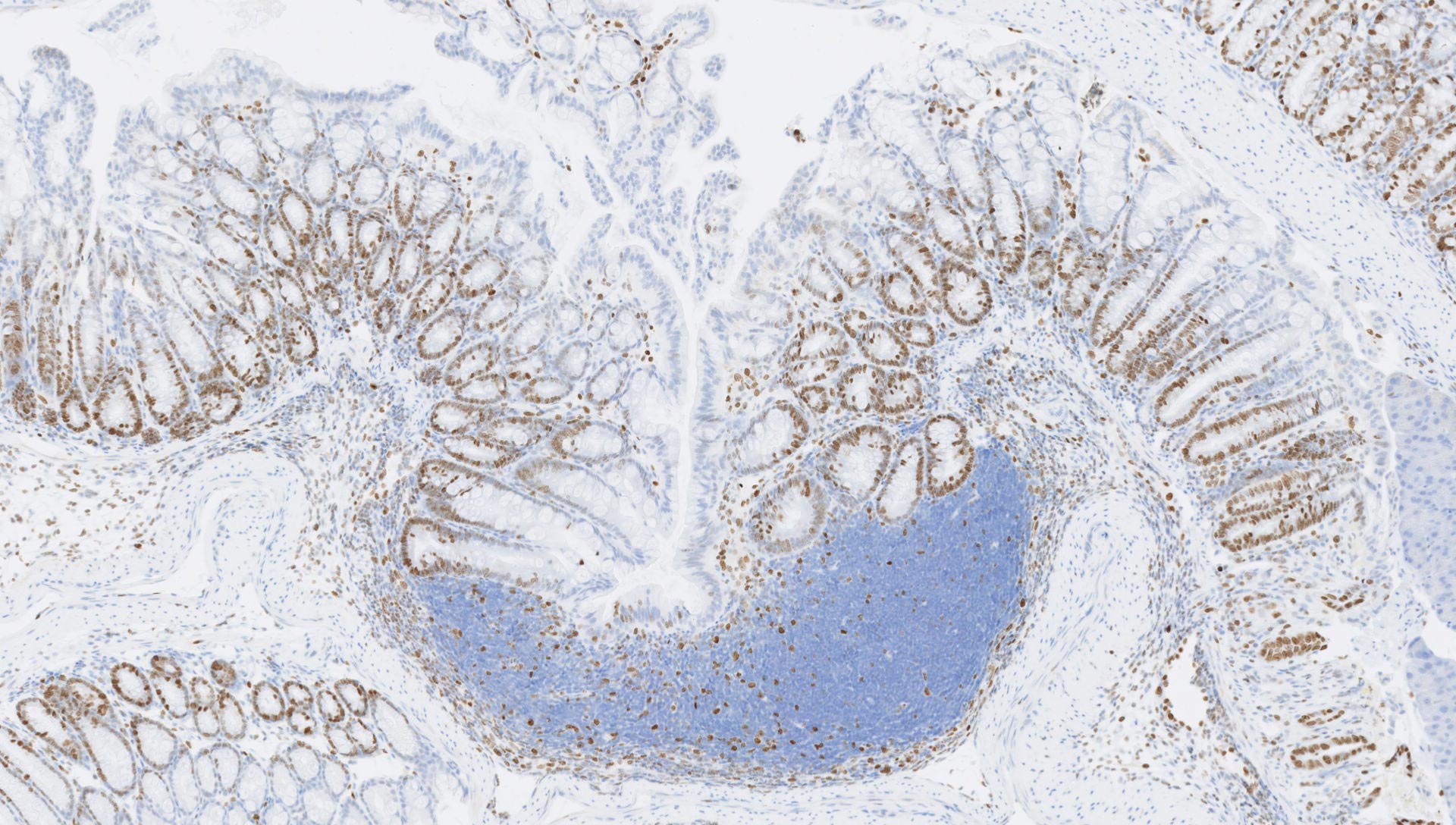
Our Histology team doesn’t just evaluate the outputs from toxicology studies. We also help evaluate structural changes, inflammatory responses, cell abnormalities in disease models and interactions between living tissue and medical devices.
We are committed to supporting your drug development journey from start to finish.
Take the next step in your preclinical drug development process and contact us today to learn more about how HistologiX can be your reliable partner in ensuring the safety and success of your pharmaceutical products. Learn more about our full range of Histology services here.